Carnet de bac
Exercices
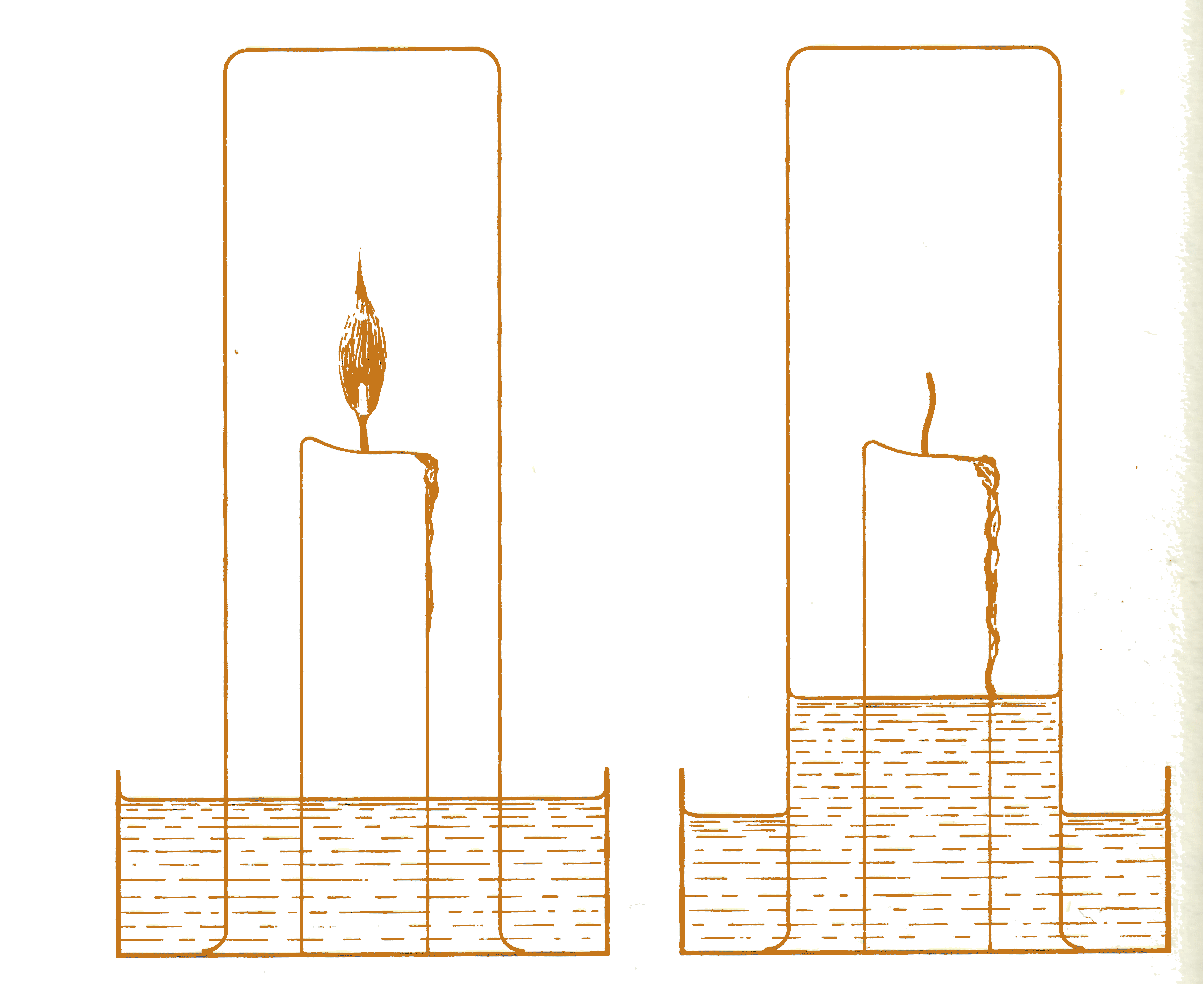
Expérience de John Mayow |
➔ |
|
Richard S. Westfall The Construction of Modern Science, 1971 Mechanical Chemistry
The work of John Mayow (1640-1679), an English doctor and chemist, further illustrattes how easily the mechanical philosophy could function to sustain a traditional point of view in chemistry. Mayow was one of a number of experimenters interested in the analogies of respiration and combustion. It was known that when a candle is burned in a closed container over water, water rises in the container and the volume of air decreases as the candle burns out. Experiments now determined that the phenomenon (in about the same quantity) occurs when a small animal expires in a simialr closed container. Not only did the reduction in volume suggest the something is removed from the air, but experiments with the air pump supported the conclusion by showing that the presence of air is necessary for both combustion and life. To these known experiments Mayow added another. He enclosed a small animal in a jar together with some combustible material that could be ignited by burning glass. When the animal had expired, the combustible material could not be set on fire ; therefore both respiration and combustion require the same substance in the air. Mayow referred to it as the nitro-aerial spirit, a name deriving from nitre (salpetre) ans expressing the fact that materials made from nitre, such a gunpowder, which contain their own mitro-aerial spirit, can burn without the presence of air.
|